| |
Modeling
stimulus transformations in extrastriate cortex
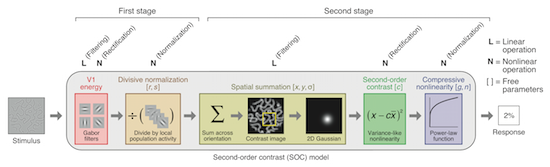
What are the stimulus computations
that are performed by extrastriate visual areas?
To answer this question, we seek to develop
quantitative computational models that predict
physiological responses in human visual cortex to
a wide range of stimuli. Our approach tightly
integrates experimental and modeling techniques
and exploits the wide coverage of visual areas
provided by fMRI. The models we develop can be
used to help understand the different types of
information encoded in different physiological
measures (e.g. single-unit responses, BOLD,
EEG/MEG, ECoG).
Selected publications
- Wandell, B.A., Winawer,
J., & Kay, K.N. Computational modeling of
responses in human visual cortex. In: Brain
Mapping: An Encyclopedic Reference, edited by P.
Thompson & K. Friston (2015). Journal
link | PDF
- Winawer, J., Kay, K.N.,
Foster, B.L., Rauschecker, A.M., Parvizi, J.,
& Wandell, B.A. Asynchronous Broadband
Signals Are the Principal Source of the BOLD
Response in Human Visual Cortex. Current
Biology 23, 1–9 (2013). Pubmed
| PDF
- Kay, K.N., Winawer, J.,
Rokem, A., Mezer, A., & Wandell, B.A. A
Two-Stage Cascade Model of BOLD Responses in
Human Visual Cortex. PLoS Computational
Biology 9(5), 1–16 (2013). Pubmed
| PDF
- Kay, K.N., Winawer, J.,
Mezer, A., & Wandell, B.A. Compressive
spatial summation in human visual cortex. Journal
of Neurophysiology 110(2), 481–493
(2013). Pubmed
| PDF
- Kay, K.N., Naselaris, T.,
Prenger, R.J., & Gallant, J.L. Identifying
natural images from human brain activity. Nature
452, 352–355 (2008). Pubmed
| PDF
Modeling
bottom-up and top-down influences on high-level
visual cortex
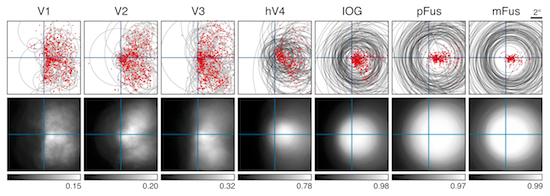
We have recently been exploring
the use of a model-based approach to understand
responses in high-level visual cortex (e.g.
face-selective and word-selective regions of
ventral temporal cortex). We seek to carefully
characterize bottom-up stimulus effects and
dissociate these from top-down cognitive
influences. These investigations are aimed towards
furthering our understanding of the relationship
between early and late visual processing and
clarifying the computational role that cognitive
processes play in visual processing.
Selected publications
- Grill-Spector,
K., Weiner, K.S., Kay, K., & Gomez, J. The
Functional Neuroanatomy of Human Face
Perception. Annual Review of Vision Science
(2017). Pubmed
- Kay, K.N. & Yeatman, J.D.
Bottom-up and top-down computations in word- and
face-selective cortex. eLife (2017). eLife | PDF
- Kay, K.N., Weiner, K.S., &
Grill-Spector, K. Attention reduces spatial
uncertainty in human ventral temporal cortex. Current
Biology (2015). Pubmed
| PDF
Population receptive field
mapping methods
 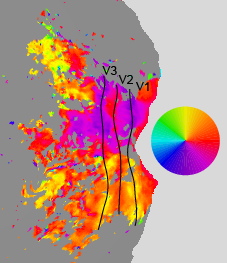
Visual space is systematically
represented in visual cortex (e.g. retinotopy).
One of the major advances of fMRI has been the
development of techniques that can be used to map the retinotopic
organization of visual cortex. Newer
techniques have been proposed to estimate
population receptive field size. We are
generally interested in developing increasingly
accurate and efficient methods for mapping
receptive fields (see analyzePRF).
Selected publications
- [NEW]
Benson, N.C., Jamison, K.W., Arcaro, M.J., Vu,
A.T., Glasser, M.F., Coalson, T.S., Van Essen,
D.C., Yacoub, E., Ugurbil, K., Winawer, J.*,
& Kay, K.* The Human Connectome Project 7
Tesla retinotopy dataset: Description and
population receptive field analysis. Journal
of Vision (2018). Pubmed
- Kay, K.N., Winawer, J., Mezer,
A., & Wandell, B.A. Compressive spatial
summation in human visual cortex. Journal
of Neurophysiology 110(2), 481–493
(2013). Pubmed
| PDF
- Kay, K.N., Naselaris, T.,
Prenger, R.J., & Gallant, J.L. Identifying
natural images from human brain activity. Nature
452, 352–355 (2008). Pubmed
| PDF
- Hansen, K.A., Kay, K.N., &
Gallant, J.L. Topographic organization in and
near human visual area V4. J. Neurosci.
27, 11896–11911 (2007). Pubmed
| PDF
|
|
Encoding and decoding approaches
to fMRI
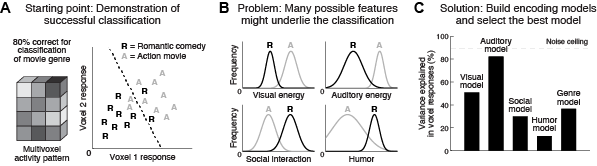
Functional magnetic resonance
imaging provides a rich source of information
about ongoing physiological activity in the human
brain. The data are highly multivariate (thousands
of distinct spatial measurements) and therefore
require effective and tractable experimental and
analysis approaches. Encoding and decoding are
useful tools for interpreting the distributed
patterns of activity measured from the brain. We
are generally interested in developing novel
analysis techniques that can help us understand
the information-processing operations performed by
the brain.
Selected publications
- Kay, K.N. Principles for models of neural
information processing. NeuroImage
(2017). Pubmed
- Naselaris, T. & Kay, K.N. Resolving
ambiguities of MVPA using explicit models of
representation. Trends in Cognitive
Sciences (2015). Pubmed
| PDF
- Kay, K.N. Understanding visual representation
by developing receptive-field models. In: Visual
Population Codes, edited by N. Kriegeskorte
& G. Kreiman (2011). Book
Link | PDF
- Naselaris, T., Kay, K.N., Nishimoto, S., &
Gallant, J.L. Encoding and decoding in fMRI. NeuroImage
56, 400–410 (2011). Pubmed
| PDF
- Kay, K.N. & Gallant, J.L. I can see what
you see. Nature Neuroscience 12,
245–246 (2009). Pubmed
| PDF
- Naselaris, T., Prenger, R.J., Kay, K.N.,
Oliver, M., & Gallant, J.L. Bayesian
reconstruction of natural images from human
brain activity. Neuron 63, 902–915
(2009). Pubmed
| PDF
- Kay, K.N., Naselaris, T., Prenger, R.J., &
Gallant, J.L. Identifying natural images from
human brain activity. Nature 452,
352–355 (2008). Pubmed
| PDF
Denoising and fMRI analysis
methods
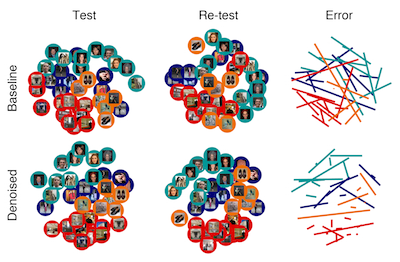
Functional magnetic resonance
imaging data suffer from numerous artifacts that
reduce signal-to-noise ratio. Given that the
experimental paradigms we use require a large
number of conditions and high signal-to-noise
ratio, we are interested in methods that can
improve data quality. Whereas some optimizations
occur at the data acquisition level, other
benefits can come in data analysis. We develop
analysis techniques that can be used to denoise
fMRI data. We are especially interested in
techniques that are practical for actual
experimental studies, and we make our software
code freely available (see GLMdenoise).
Selected publications
- [NEW]
Charest, I., Kriegeskorte, N., & Kay, K.
GLMdenoise improves multivariate pattern
analysis of fMRI data. NeuroImage
(2018). Pubmed
- Henriksson, L.,
Khaligh-Razavi, S., Kay, K., & Kriegeskorte,
N. Visual representations are dominated by
intrinsic fluctuations correlated between areas.
NeuroImage (2015). Pubmed
| PDF
- Kay, K.N., Rokem, A., Winawer,
J., Dougherty, R.F., & Wandell, B.A.
GLMdenoise: a fast, automated technique for
denoising task-based fMRI data. Frontiers
in Neuroscience 7 (2013). Pubmed
| PDF
- Kay, K.N., David, S.V.,
Prenger, R.J., Hansen, K.A., & Gallant, J.L.
Modeling low-frequency fluctuation and
hemodynamic response timecourse in event-related
fMRI. Hum. Brain Mapp. 29, 142–156
(2008). Pubmed
| PDF
|
|





